
Protests over Israel-Hamas war lead to hundreds of arrests on campus
Hundreds of people have been arrested in California, New York, Massachusetts, Texas, Georgia and other states during the tense protests on college campuses.
Watch CBS News

Hundreds of people have been arrested in California, New York, Massachusetts, Texas, Georgia and other states during the tense protests on college campuses.

The Supreme Court convened to consider whether former President Donald Trump is entitled to broad immunity from criminal charges in the 2020 election case.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Former National Enquirer boss David Pecker appeared on the stand for the third day, detailing an agreement the tabloid made with a former Playboy model.

A former high school athletic director was arrested Thursday morning after allegedly using artificial intelligence to impersonate the school principal in a recording that included racist and antisemitic comments.

A federal judge has denied former President Donald Trump's request for a new trial in the civil suit brought by the writer E. Jean Carroll, who was awarded an $83.3 million judgment.

The petitions are the latest in the effort for Ryan Corbett's release.

President Joe Biden has signed legislation that could lead to TikTok being sold or banned. Here's who might buy it — and for how much.

U.S. regulators are reviving a rescinded rule, laying the groundwork for for a major court fight with the broadband industry.

Astronauts Barry Wilmore and Sunita Williams say they have complete confidence in the Starliner despite questions about Boeing's safety culture.

David Schultz's wife said that the person found was wearing boots that matched her husband's, and his keys were found in the pants pocket.

Senate Minority Leader Mitch McConnell appears on "Face the Nation" as pro-Palestinian protests roil American politics.

A cross unearthed in eastern Poland likely belonged to an outcasted Russian religious community around 300 years ago.

The median mortgage payment jumped to a record $2,843 in April, up nearly 13% from a year ago, a new analysis finds.

Henderson police said they found the boy wearing a diaper in the feces-smeared enclosure.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

A photo of the raft the man is believed to have used shows just a few long planks assembled together.

The Shenzhou 18 crew will replace three taikonauts aboard the Chinese space station who are wrapping up a six-month stay.

There are no cameras allowed in the court where Trump is being tried on 34 felony counts stemming from a "hush money" payment before the 2016 election.

Jurors in former President Donald Trump's trial in New York heard testimony from a former media executive about his efforts to bury negative stories about Trump before the 2016 presidential election.

Trump made 10 social media posts that were "threatening, inflammatory," prosecutors said, arguing he should pay a fine for each post.

The FBI calls on tech companies to "step up" to protect people looking for love online.

Laura Kowal's match on an online dating site wasn't what he seemed. Now her daughter is on a mission to expose the risk of romance scams: "It could happen to anybody."

Scammers have been increasingly successful in leveraging their romantic grip on victims.

Summer Lee has defeated Bhavini Patel in the Democratic primary for Pennsylvania's 12th Congressional District, the Associated Press projects.

He could receive a large windfall from his newly public media company, Trump Media & Technology Group.

Protesters have been arrested at Columbia and Yale as they've called for a break from Israel.

As Israel's leader equates U.S. university protests to rallies in Nazi Germany, Palestinian students tell CBS News what the support means to them.

House Speaker Mike Johnson was met with loud boos as he visited Columbia University, where he joined calls for the president's resignation amid pro-Palestinian protests.
The video appears to show U.S.-Israeli hostage Hersh Goldberg-Polin delivering a message under duress.

Do you have $7,500 or more in credit card debt? Here are a few ways to pay it off quickly.

Looking to make a successful investment in gold? Then be sure to avoid making these simple mistakes.

You don't have to accept a low interest rate on your savings. There are many great account options to consider.

Visitors will have to pay five euros, a fee designed to offset some of the costs of accommodating tourists.

President Joe Biden has signed legislation that could lead to TikTok being sold or banned. Here's who might buy it — and for how much.

These are the airports Southwest is pulling out of completely as it looks to save costs.

Only one vehicle of the 10 small SUVs tested earned a good rating.

Some 46.8% of luxury homes were bought entirely with cash in the three months ended February 29, the highest share in a decade, according to Redfin.

Receive a $40 Digital Costco Shop Card when you join as a new member at Costco.com when entering PARA24 at checkout.

Using one of the best outdoor projector is the perfect way to host a movie night for friends and family.

Save big on each model of Apple AirPods ahead of the holiday by shopping our curated selection of deals.

A New York appeals court overturned Harvey Weinstein's 2020 conviction on felony sex crimes. The court ruled that the disgraced movie mogul did not have a fair trial because the judge who presided over the case allowed women to testify about allegations that were not part of the charges against him. Weinstein will remain in prison because of his rape conviction in Los Angeles.

The Supreme Court has concluded arguments over Donald Trump's claim he is entitled to broad immunity from federal prosecution for allegedly official acts while he was in the White House. Nancy Cordes anchored CBS News' special report on the arguments.

A fourth temporary channel is opening for the Port of Baltimore one month after the Francis Scott Key Bridge collapsed. Two victims of the collapse still have not been recovered as crews continue to remove the wreckage. Col. Estee Pinchasin, Baltimore District commander for the U.S. Army Corps of Engineers, joined CBS News to discuss the recovery.

Another tense day of protests over the Israel-Hamas war is expected on college campuses across the country on Thursday. Hundreds of people have already been arrested since the demonstrations began. CBS News Boston reporter Penny Kmitt reports.

A surfing accident left New York teacher Billy Keenan paralyzed, but when he received a call from a police officer, his life changed.

A mom was worried about what her son, who has autism, would do after high school. So she opened the So Much To Give cafe, a restaurant in Cedars, Pennsylvania, that employs people with disabilities – and helps them grow.

David Begnaud visits Jeffrey Olsen, known as the "Toy Man" in Vista, California, who has dedicated over 30 years to donating toys, food, and clothes to those in need.

David Begnaud has the story of an unlikely friendship at a Massachusetts elementary school, proving that words are not always necessary to form a special connection.

A bus driver in Milwaukee, Wisconsin, sprang into action when he saw a toddler on the street barefoot and wandering alone. The driver rescued the child from the cold and then contacted authorities.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

In an exclusive interview with "CBS Evening News" anchor and managing editor Norah O’Donnell, Pope Francis discusses the wars in Ukraine and Gaza and calls for negotiating peace.

President Biden and his reelection campaign clearly state they need Latino support this November. However, several polls in recent months have revealed an increasing number of Latino voters preferring former President Donald Trump. CBS News political director Fin Gómez and Democratic strategist Chuck Rocha join to discuss more.

"CBS Evening News" anchor and managing editor Norah O'Donnell was given a private tour of the magnificent St. Peter's Basilica, the largest church in the world, located in the smallest nation in the world.

CBS News asked Catholics in cities across the U.S. about their relationship with the church and how they feel about the pope. Nikki Battiste has more.

The Supreme Court heard oral arguments Wednesday over an Idaho abortion law that could have an impact on emergency room care in that state and beyond. CBS News legal correspondent Jan Crawford unpacks the proceedings.

House Speaker Mike Johnson visited Columbia University on Wednesday as pro-Palestinian protests continued on the campus. CBS News' Nancy Chen spoke with the speaker about the demonstrations.

Arizona lawmakers moved forward Wednesday in their efforts to repeal a Civil War-era law that bans all abortions except to save the woman's life. CBS News campaign reporter Shawna Mizelle has the details.

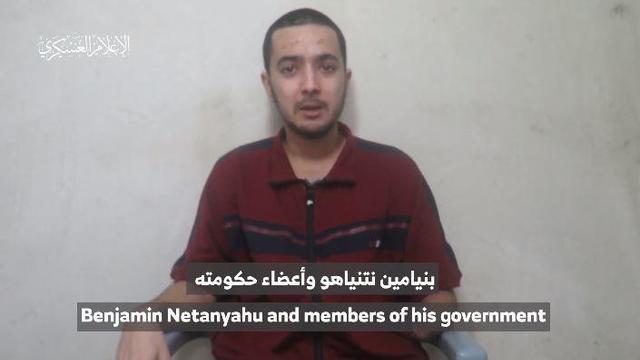
A U.S. official confirmed to CBS News that there are five American hostages believed to be alive and held by Hamas. Hamas released a propaganda video Wednesday that appears to show U.S.-Israeli hostage Hersh Goldberg-Polin. CBS News chief foreign affairs correspondent and "Face the Nation" moderator Margaret Brennan joins with more.

Former United Nations Ambassador Nikki Haley won more than 16% of the vote in Pennsylvania's Republican primary on Tuesday despite no longer being in the 2024 race. Anthony Salvanto, Matt Gorman and Caitlin Huey-Burns join "America Decides" to assess what it could mean for former President Donald Trump.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

At his lowest moment, U.S. Army veteran and former teacher Billy Keenan found strength in his faith as he was reminded of his own resilience.

A surfing accident left New York teacher Billy Keenan paralyzed, but when he received a call from a police officer, his life changed.

The So Much To Give Inclusive Cafe in Cedars, Pennsylvania employs 63 people — 80% have a disability.

A mom was worried about what her son, who has autism, would do after high school. So she opened the So Much To Give cafe, a restaurant in Cedars, Pennsylvania, that employs people with disabilities – and helps them grow.

CBS Reports goes to Illinois, which has one of the highest rates of institutionalization in the country, to understand the challenges families face keeping their developmentally disabled loved ones at home.

As more states legalize gambling, online sportsbooks have spent billions courting the next generation of bettors. And now, as mobile apps offer 24/7 access to placing wagers, addiction groups say more young people are seeking help than ever before. CBS Reports explores what experts say is a hidden epidemic lurking behind a sports betting bonanza that's leaving a trail of broken lives.

In February 2023, a quiet community in Ohio was blindsided by disaster when a train derailed and authorities decided to unleash a plume of toxic smoke in an attempt to avoid an explosion. Days later, residents and the media thought the story was over, but in fact it was just beginning. What unfolded in East Palestine is a cautionary tale for every town and city in America.

In the aftermath of the Supreme Court striking down affirmative action in college admissions, CBS Reports examines the fog of uncertainty for students and administrators who say the decision threatens to unravel decades of progress.

CBS Reports examines the legacy of the U.S. government's terrorist watchlist, 20 years after its inception. In the years since 9/11, the database has grown exponentially to target an estimated 2 million people, while those who believe they were wrongfully added are struggling to clear their names.

Astronauts Barry Wilmore and Sunita Williams say they have complete confidence in the Starliner despite questions about Boeing's safety culture.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

Some 46.8% of luxury homes were bought entirely with cash in the three months ended February 29, the highest share in a decade, according to Redfin.

The median mortgage payment jumped to a record $2,843 in April, up nearly 13% from a year ago, a new analysis finds.

Former National Enquirer boss David Pecker appeared on the stand for the third day, detailing an agreement the tabloid made with a former Playboy model.

Some 46.8% of luxury homes were bought entirely with cash in the three months ended February 29, the highest share in a decade, according to Redfin.

The median mortgage payment jumped to a record $2,843 in April, up nearly 13% from a year ago, a new analysis finds.

U.S. regulators are reviving a rescinded rule, laying the groundwork for for a major court fight with the broadband industry.

Visitors will have to pay five euros, a fee designed to offset some of the costs of accommodating tourists.

PayPal payments are being made to 117,044 consumers whose videos may have been accessed by unauthorized users.

Senate Minority Leader Mitch McConnell appears on "Face the Nation" as pro-Palestinian protests roil American politics.

Former National Enquirer boss David Pecker appeared on the stand for the third day, detailing an agreement the tabloid made with a former Playboy model.

The petitions are the latest in the effort for Ryan Corbett's release.

A federal judge has denied former President Donald Trump's request for a new trial in the civil suit brought by the writer E. Jean Carroll, who was awarded an $83.3 million judgment.

Coal-fired power plants would have to capture smokestack emissions or shut down under a new EPA rule the industry says would make the grid less reliable. It's likely to face court challenges.

CDC's provisional figures show a 2% decline in births from 2022 to 2023.
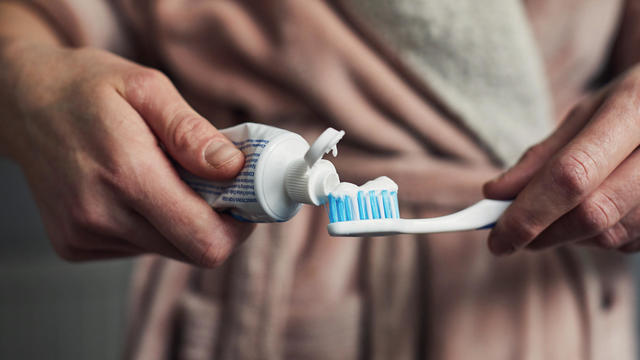
Don't brush your teeth after breakfast? Or after vomiting? Dentists say it can wear away your enamel. Here's what to do instead.

Federal officials say they're double checking whether pasteurization has eradicated the danger from possible bird virus particles in milk.
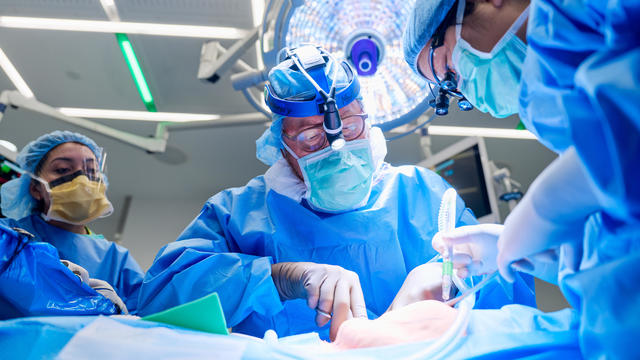
For the first time, surgeons at NYU Langone Health performed a combined mechanical heart pump and gene-edited pig kidney transplant into a living person.

The USDA had floated banning flavored milk options from some school lunches.

The petitions are the latest in the effort for Ryan Corbett's release.

A cross unearthed in eastern Poland likely belonged to an outcasted Russian religious community around 300 years ago.

Hundreds of people have been arrested in California, New York, Massachusetts, Texas, Georgia and other states during the tense protests on college campuses.
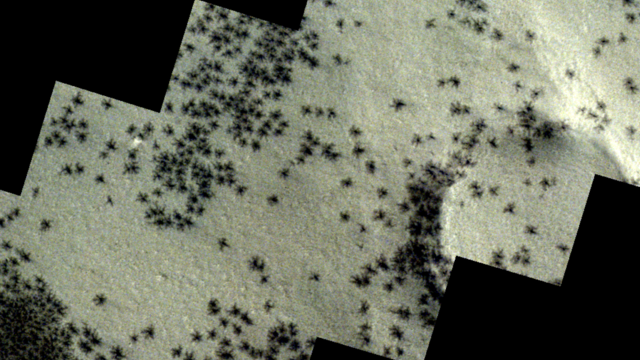
The creepy patterns were observed by the European Space Agency's ExoMars Trace Gas Orbiter.

The Shenzhou 18 crew will replace three taikonauts aboard the Chinese space station who are wrapping up a six-month stay.

Looking for a place to live in NYC? Zillow is now listing Frank Sinatra and Mia Farrow's former home on the Upper East Side.

Italy's Culture Ministry has banned loans of works to the Minneapolis Institute of Art, following a dispute with the U.S. museum over an ancient marble statue believed to have been looted from Italy almost a half-century ago.

The renowned Moulin Rouge cabaret venue's director has vowed to "rise to the challenge" after the windmill's sails fell off.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

Taylor Swift fans have found a way to feel "a little bit closer to" their hero at a London watering hole, and The Black Dog pub is lapping it up.

U.S. regulators are reviving a rescinded rule, laying the groundwork for for a major court fight with the broadband industry.

Meta began rolling out its new AI-powered smart assistant software, saying it will be integrated across Instagram, Facebook and Messenger. Adam Auriemma, editor-in-chief for CNET, joined CBS News to discuss the new tool.

From labor shortages to environmental impacts, farmers are looking to AI to help revolutionize the agriculture industry. One California startup, Farm-ng, is tapping into the power of AI and robotics to perform a wide range of tasks, including seeding, weeding and harvesting.

Lawmakers argue the Chinese government can use the widely popular video-sharing app as a spy tool and to covertly influence the U.S. public.

NASA's Voyager 1, the first spacecraft to travel beyond our solar system, has started sending information back to Earth again after scientists managed to fix the probe from 15 billion miles away.

Emerging cicadas are so loud in one South Carolina county that residents are calling the sheriff's office asking why they can hear a "noise in the air that sounds like a siren, or a whine, or a roar." CBS News' John Dickerson has details.

Representatives from across the world are gathering in Ottawa, Canada, to negotiate a potential treaty to limit plastic pollution. CBS News national environmental correspondent David Schechter has the latest on the talks.

"Although to some, the noise is annoying, they pose no danger to humans or pets," the sheriff wrote. "Unfortunately, it is the sounds of nature."

The White House is considering declaring a national climate emergency to unlock federal powers and stifle oil development, according to a Bloomberg report. Meanwhile, the Biden administration is announcing several projects this Earth Week. Columbia University Climate School professor Dr. Melissa Lott joins with analysis.

NASA's Voyager 1, the first spacecraft to travel beyond our solar system, has started sending information back to Earth again after scientists managed to fix the probe from 15 billion miles away.

The State of New York Court of Appeals overturned Harvey Weinstein's 2020 rape conviction Thursday and has ordered a new trial. Julie Rendelman, a criminal defense attorney, and CBS News national correspondent Jericka Duncan look at the possible reasons why it was overturned and what it means for Weinstein, who was also convicted of rape in Los Angeles in 2022.

A New York appeals court overturned Harvey Weinstein's 2020 conviction on felony sex crimes. The court ruled that the disgraced movie mogul did not have a fair trial because the judge who presided over the case allowed women to testify about allegations that were not part of the charges against him. Weinstein will remain in prison because of his rape conviction in Los Angeles.

Harvey Weinstein's 2020 conviction on felony sex crime charges has been overturned by the State of New York Court of Appeals.

A former high school athletic director was arrested Thursday morning after allegedly using artificial intelligence to impersonate the school principal in a recording that included racist and antisemitic comments.

William Ray Grimes was indicted on charges of murder and burglary in the 2012 slaying of Lowell Badger, police said.

Astronauts Barry Wilmore and Sunita Williams say they have complete confidence in the Starliner despite questions about Boeing's safety culture.

In 1961, Ed Dwight was selected by President John F. Kennedy to enter an Air Force training program known as the path to NASA's Astronaut Corps. But he ultimately never made it to space.

The creepy patterns were observed by the European Space Agency's ExoMars Trace Gas Orbiter.

The Shenzhou 18 crew will replace three taikonauts aboard the Chinese space station who are wrapping up a six-month stay.

In November 2023, NASA's Voyager 1 spacecraft stopped sending "readable science and engineering data."

A look back at the esteemed personalities who've left us this year, who'd touched us with their innovation, creativity and humanity.

The Francis Scott Key Bridge in Baltimore collapsed early Tuesday, March 26 after a column was struck by a container ship that reportedly lost power, sending vehicles and people into the Patapsco River.

When Tiffiney Crawford was found dead inside her van, authorities believed she might have taken her own life. But could she shoot herself twice in the head with her non-dominant hand?

We look back at the life and career of the longtime host of "Sunday Morning," and "one of the most enduring and most endearing" people in broadcasting.

Cayley Mandadi's mother and stepfather go to extreme lengths to prove her death was no accident.

An Arizona grand jury has indicted 18 people, including several allies of former President Trump such as Rudy Giuliani and Mark Meadows, over an alleged attempt to alter the results of the 2020 presidential election. CBS News' Shawna Mizelle reports.

Testimony continued Thursday in former President Donald Trump's criminal "hush money" trial. David Pecker, the former publisher of the National Enquirer, detailed alleged efforts to suppress stories involving adult film star Stormy Daniels. CBS News' Errol Barnett reports.

The 2024 NFL Draft kicks off in Detroit on Thursday, and all eyes will be on USC quarterback Caleb Williams, who is expected to be the first overall draft pick. NFL on CBS reporter Aditi Kinkhabwala joins CBS News with more.

Police and protesters are clashing at U.S. college campuses as fears of antisemitism and Islamophobia grow among students and faculty members. Gelila Negesse, a senior staff writer for the Columbia Daily Spectator, joins CBS News with more.

A new video released by Hamas appears to show Hersh Goldberg-Polin, an Israeli-American hostage taken during the Oct. 7 attacks in Israel. His mother, Rachel Goldberg-Polin, tells CBS News' Debora Patta what it was like hearing her son's voice.